Antibiotic resistance is slowly becoming one of the greatest threats to public health worldwide, as it renders existing treatments ineffective against constantly adapting bacteria. While searching for methods to combat the waning power of antibiotic treatments, scientists have discovered an answer within our own immune systems.
Our bodies produce natural antibodies to thwart harmful bacteria and viruses as the first line of defense, and researchers have found a way to repurpose and strengthen these antibodies to fight the battles that antibiotics cannot. They were able to develop tissue-specific gene therapy using lysozyme (LYZ) and tracheal antimicrobial peptide (TAP)—two natural antibacterial molecules—that has the potential to treat patients with bacterial infections in a safer and more sustainable way. This gene-based therapy, tested in mouse models with mastitis—an infection causing inflammation in the breasts—shows the therapy’s ability to perform against both Gram-positive and Gram-negative bacteria, offering an innovative and potentially game-changing alternative to traditional antibiotics.
The Antibiotic Problem

The overuse of antibiotics in both human and veterinary medicine has led to the widespread rise of resistant bacterial strains, making formerly treatable infections deadly and costly. In the U.S. alone, over 23,000 people die each year from resistant infections, with billions spent on healthcare and lost productivity. In the agricultural sector, millions of pounds of antibiotics are administered annually to livestock, contributing significantly to environmental and food chain contamination. The race to find antibiotic-free solutions has intensified, and LYZ/TAP-based gene therapy may be leading the pack.
Unlike conventional antibiotics, LYZ and TAP are innate defense molecules that animals already produce to fight infection. Lysozyme works by breaking down bacterial cell walls, while TAP disrupts bacterial membranes through ionic interference. These molecules are naturally potent against a wide array of pathogens and are unlikely to induce resistance due to their mechanisms of action.
Study Parameters
Though the study’s conclusions provide us with a wide range of uses, researchers solely focused on developing tissue-specific expression antimicrobial plasmids (TSEAP) that target the mammary gland for this study’s purpose. Researchers used a special promoter (β-lactoglobulin gene) to ensure that LYZ and TAP are only expressed in the intended tissue, minimizing side effects elsewhere in the body.
Using a non-infected carrier, the plasmids were successfully transfected into mouse mammary glands, which then began producing LYZ or TAP proteins for up to 48 hours. The transfection process was localized, efficient, and safe. Mice treated with these plasmids showed strong expression of the therapeutic proteins without systemic toxicity, as confirmed through blood biochemistry, organ weight analysis, and behavioral tests. Unlike traditional antibiotics, which affect the whole body and often the surrounding environment, this method acts like a smart bomb, delivering the therapeutic precisely where it’s needed.
Testing Efficacy
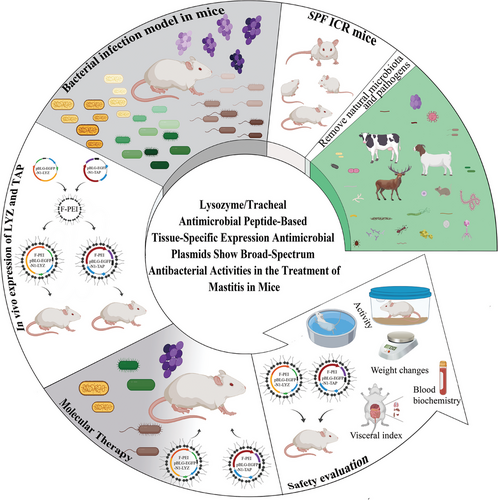
To test the treatment’s efficacy, researchers administered the gene-based therapy to mice infected with four common mastitis-causing bacteria: Staphylococcus sciuri, Bacillus cereus, Escherichia coli, and Pseudomonas aeruginosa. The study yielded promising results across the board. Mice receiving LYZ/TAP therapy showed reduced body temperatures, decreased inflammatory cytokines (such as IL-1β, IL-6, and TNF-α), and less histopathological damage compared to untreated mice. In some cases, symptoms disappeared almost entirely, especially in mice infected with B. cereus and treated with LYZ.
Moreover, the therapy not only halted the infection but also reversed immune dysregulation. White blood cell counts returned to near-normal levels, and inflammation-related damage in the mammary tissue was significantly reduced. Even in cases where traditional treatment might have required multiple rounds of antibiotics, a single transfection appeared sufficient to confer therapeutic benefit.
Future Prospects
Although the research was conducted in mice, the implications of the findings are vast. The therapeutic approach could one day treat mastitis in dairy cows, reducing antibiotic use and preventing resistant bacteria from entering the food supply. Furthermore, with appropriate tissue-specific promoters, this method could be adapted to other organs and diseases, potentially reshaping how bacterial infections are treated in humans and animals.
However, scientists caution that there is still a long way to go and many challenges to overcome before therapy-based treatments can address the vast array of bacterial infections. One significant obstacle is the limited duration of expression, lasting less than two days, while the cost of gene-based therapy remains high. Long-term immune response and large-scale manufacturing need addressing before such therapies can become mainstream.
Nevertheless, LYZ/TAP-based plasmid therapy represents a bold step forward in combating antibiotic resistance. By harnessing our natural defenses and delivering them with surgical precision, this research opens the door to a future where antibiotics are no longer the sole defense against bacterial diseases.
